Children’s National Advances Pediatric Brain Tumor Research with NaviFUS Focused Ultrasound Platform

Children’s National Hospital is advancing new possibilities in pediatric brain tumor care through expanded focused ultrasound research using the NaviFUS neuronavigation-guided low-intensity focused ultrasound (LIFU) platform. The collaboration supports ongoing efforts to explore non-invasive therapeutic approaches for children with difficult-to-treat brain tumors.
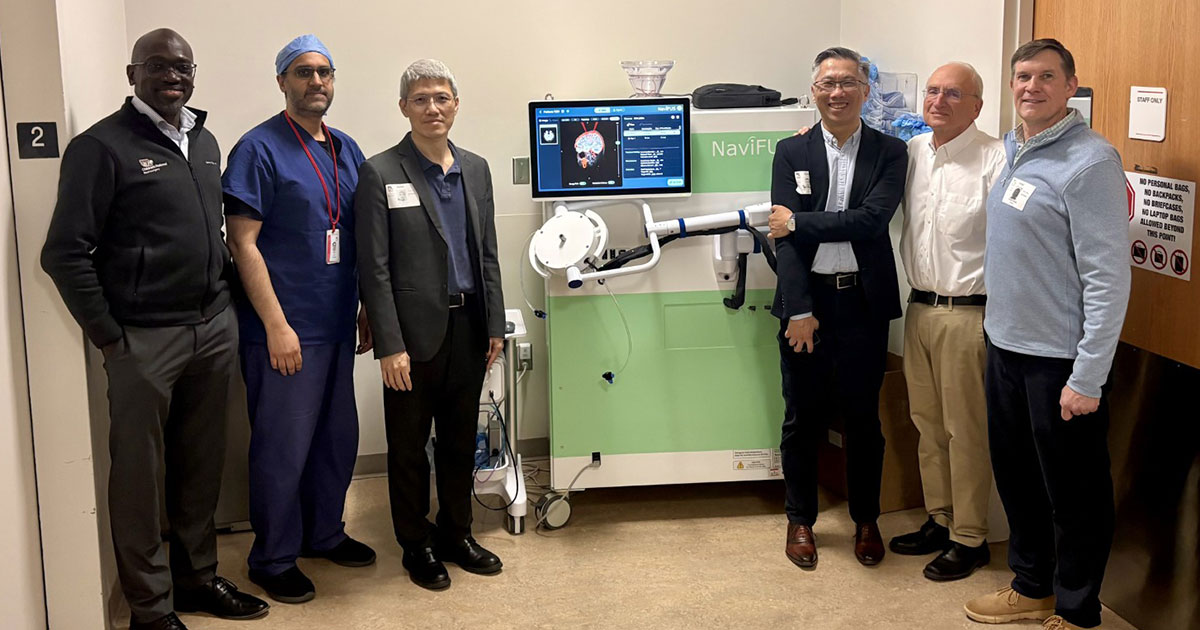
Children’s National physician-scientists Chima Oluigbo, MD, Hasan Syed, MD, and Roger J. Packer, MD, welcomed the new NaviFUS focused ultrasound system alongside NaviFUS Corp. founder Hao‑Li Liu, PhD, Chief Executive Officer Arthur Lung, PhD, and Clinical Applications Manager David Moore
Children’s National Hospital is advancing brain tumor care and research with a second-generation low-intensity focused ultrasound (LIFU) system donated by Taiwan-based NaviFUS Corp. This platform builds on existing capabilities and will support the development of more precise, less invasive treatments for children with brain tumors.
The system will expand clinical trials testing new therapies, including LIFU-based approaches. With two advanced systems and active first-in-human studies, Children’s National is now the only freestanding pediatric hospital in the world with this level of focused ultrasound capability dedicated to pediatric brain tumor research.
The big picture
Many childhood brain tumors are in areas where surgery is not possible, or medicines can’t reach. This makes drug delivery a major challenge. LIFU is a noninvasive technology under clinical investigation that can temporarily open the protective blood-brain barrier to help therapies reach tumors that are otherwise difficult or impossible to treat.
Children’s National is a leader in LIFU research, with an integrated program that moves discoveries from the laboratory into clinical trials. Preclinical studies in partnership with Virginia Tech explore whether LIFU can enhance the delivery of emerging immunotherapies, such as CAR-T and T-cell therapies. Clinically, investigators are leading first-in-human pediatric trials using LIFU to safely open the blood-brain barrier and deliver targeted treatments. Early results show strong safety and encouraging signs of benefit in children with high-grade gliomas, including diffuse intrinsic pontine glioma (DIPG).
The donation reflects NaviFUS’s recognition of Children’s National as a leader in focused ultrasound. A recent pilot study published in Neurosurgery supports the reliability of the NaviFUS system, showing that repeated LIFU-mediated blood-brain barrier opening is safe and feasible in adults with recurrent glioblastoma (rGBM) with encouraging results7.
“Our collaboration with Children’s National represents a pivotal step in expanding the clinical reach of our technology,” says Arthur Lung, PhD, CEO of NaviFUS Corp. “By providing this second-generation system, we aim to translate our successful findings in adult rGBM into the pediatric setting, offering a safer, more effective way to breach the blood-brain barrier and deliver hope to families facing the most challenging diagnoses.”
The patient impact
The NaviFUS system brings meaningful advances for patients and families. By integrating real-time neuronavigation — a computer-assisted, image-guided technology — with focused ultrasound, it delivers highly precise treatment without the need for a fixed frame. It supports repeated treatments within standard care, reducing the complexity of intracranial drug delivery. Its approach also allows for shorter procedures and may reduce sedation requirements, making treatment easier for patients.
With two advanced focused ultrasound systems now at Children’s National, more children may have access to personalized care and clinical trial options. It also gives care teams more flexibility to tailor therapies to each child’s needs.
Moving the Field Forward
The addition of a NaviFUS system is expected to speed up clinical trials and help bring novel treatments to patients sooner. This includes a new first-in-human pediatric LIFU trial starting this summer, as well as future studies combining LIFU with immunotherapies.
Focused ultrasound may also have broader uses in pediatric neurosurgery. Its precision nature could expand treatment options for conditions that currently have limited therapies, including epilepsy, movement disorders and rare genetic diseases.
“The next step is making focused ultrasound something we can do reliably and integrate into a real workflow,” says pediatric neurosurgeon Hasan Syed, MD, who co-directs the Focused Ultrasound Program and helped bring the new system to Children’s National. “It will strengthen the foundation for brain tumor studies and position us to evaluate other focused ultrasound applications in neurosurgery, as the evidence supports it.”
Beyond functionality, the NaviFUS system offers hope to children facing hard-to-treat tumors.
“This investment reflects our commitment to advancing new therapies for patients who have limited options,” says Roger J. Packer, MD, director of the Brain Tumor Institute and the Gilbert Family Neurofibromatosis Institute. “We are grateful to donors like NaviFUS for helping us move the field forward and bring better treatments closer to the children who need them most.”
View full press release
Advancing LIFU-enabled therapies for pediatric brain tumor care May 4, 2026
